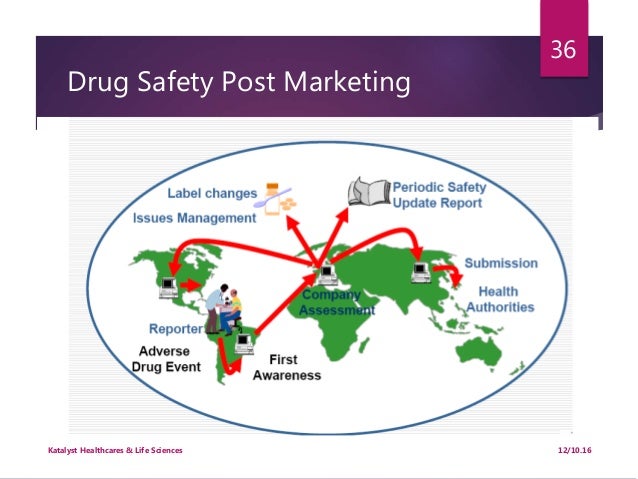
. PADERs are postmarket periodic drug safety reports required in the USA by FDA regulations §§314.80(c)(2) and 600.80(c)(2). PADERs have largely been superceded by the new Public Benefit-Risk Evaluation Report (PBRER) defined in ICH E2C(R2), following FDA endorsement in November 2012 of the format designed for harmonised use across all ICH regions. The PBRER also forms the basis of the current version of the European Periodic Safety Update Report (PSUR). PADERs/PBRERs are required to be submitted quarterly for the first 3 years after drug approval in the USA, and annually thereafter. Trilogy’s experience with PSURs extends across a wide range of indications. As a provider also of pre-approval safety documentation, CTDs and other drug approval documentation, Trilogy can also provide the 120 Day Safety Update required by the FDA immediately following CTD submission.
- Fda Periodic Safety Update Reports Download
- Fda Periodic Safety Update Reports Free
- Fda Safety Update Report Guidance
The development safety update report (DSUR) proposed in this guidance is intended to be a common standard for periodic reporting on drugs under development (including marketed drugs that are under. An Overview of the Periodic Safety Update Report for Marketed Drugs E2C (R2) Overview GVP Module VII: PSURs. The final concept paper was entitled Periodic Safety Update Reports for Marketed Drugs E2C(R2) and gap and potential improvement analysis of ICH E2C, E2E and E2F. Investigational drug exposure in healthy volunteers might be less.
Fda Periodic Safety Update Reports Download
During the drug development and after the drug is on the market, the sponsor or market authorization holder has obligations to submit the safety information to regulatory agencies periodically. These periodic reports have different requirements and sometimes are confusing.IB:According to ICH E6 “Good Clinical Practice”,The Investigator's Brochure (IB) is a compilation of the clinical and nonclinical data on the investigational product(s) that are relevant to the study of the product(s) in human subjects. Its purpose is to provide the investigators and others involved in the trial with the information to facilitate their understanding of the rationale for, and their compliance with, many key features of the protocol, such as the dose, dose frequency/interval, methods of administration, and safety monitoring procedures. The IB also provides insight to support the clinical management of the study subjects during the course of the clinical trial.For post marketing commitment clinical trials, the product label (package insert) may be used in place of the investigator brochure since the package insert includes the contents required in the investigator brochure. According to the ICH E2C (R2) “”,The Periodic Benefit-Risk Evaluation Report (PBRER) described in this Guideline is intended to be a common standard for periodic benefit-risk evaluation reporting on marketed products (including approved drugs that are under further study) among the ICH regions.Other supporting documents regarding PBRER are:.E2C (R2) is finalized at November 2012 and is supposed to replace the PSUR.


Fda Periodic Safety Update Reports Free
However, EMA has not fully adopted the PBRER and continues to use the term PSUR as defined in its recent guideline “”FDA fully endorsed E2C (R2) and PBRER and issued its guidance “”A paper from Sentrx.com discussed this confusion.PBRER vs. DSURIt is often confusing whether or not a PBRER or DSUR or both are needed. It is commonly understood that PBRER is for a marketed products (including approved drugs that are under further study) and DSUR is for drugs under development (including marketed drugs that are under further study). In US, for the marketed products, if IND is still open, the annual safety report (i.e., DSUR) will be required by FDA.There are some duplications between the DSUR and PBRER.
Fda Safety Update Report Guidance
FDA's guidance ' intended to provide some clarifications about PBRER and DSUR. In some situations, both PBRER and DSUR are required (to meet different regulatory requirements). In preparation. However, the effort can be made to minimize the duplicate works.' This guideline aims to address this duplication and facilitate flexibility by encouraging the use of individual modules, where they pertain to more than one report – to be used at different times, for different authorities, and for different purposes. Therefore, the PBRER has been developed in such a way that content of several sections may be used for sections of other documents as a basis for a modular approach (see Section 1.1). 'To some degree, the DSUR can be considered as a subset of PBRER with focus on the ongoing clinical trials.